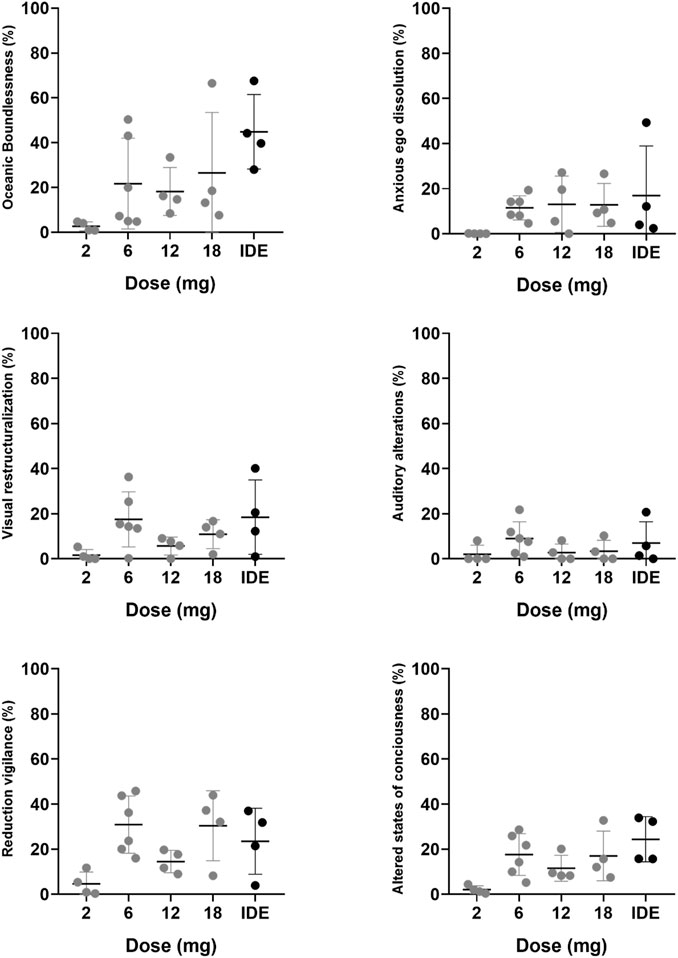
Frontiers | A Phase 1, Dose-Ranging Study to Assess Safety and Psychoactive Effects of a Vaporized 5-Methoxy-N, N-Dimethyltryptamine Formulation (GH001) in Healthy Volunteers
Com-COV vaccine study to research third dose booster options for 12-to-15-year-olds | University of Oxford

CDC on Twitter: "Children get the #COVID19 vaccine dose recommended for their age group at the time of administration. For example, if your child is 11 for their first dose & 12

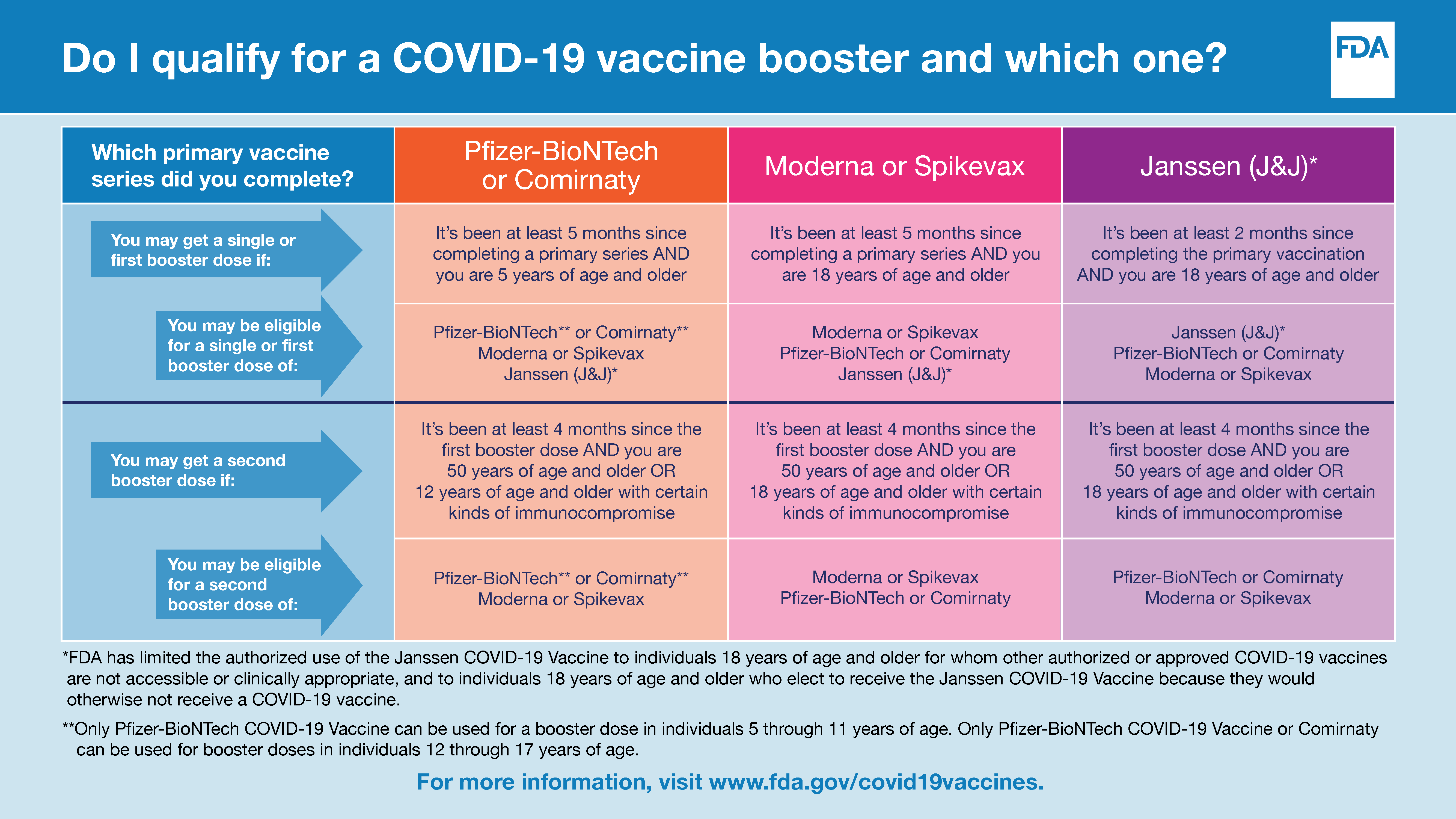
Updated Toolkit: COVID-19 Booster Dose Messaging and Outreach Tools - Public Health Communication Collaborative
Postmarketing active surveillance of myocarditis and pericarditis following vaccination with COVID-19 mRNA vaccines in persons aged 12 to 39 years in Italy: A multi-database, self-controlled case series study | PLOS Medicine

FDA authorizes new guidance on Pfizer vaccines and boosters for children and adults : Oregon Health News

COVID-19 Vaccine Second-Dose Completion and Interval Between First and Second Doses Among Vaccinated Persons — United States, December 14, 2020−February 14, 2021 | MMWR