
Hs Humira Citrate-free Adult And Adolescent Initial - Hidradenitis Suppurativa Humira Dose PNG Image | Transparent PNG Free Download on SeekPNG

Five-year analysis from the ESPRIT 10-year postmarketing surveillance registry of adalimumab treatment for moderate to severe psoriasis - Journal of the American Academy of Dermatology

Humira rings up $20.7B in 2021, but AbbVie still mum on post-biosimilar expectations | Fierce Pharma

Humira Injection Review In Hindi | Adalimumab Injection Uses, Precautions, Dose & Side Effects - YouTube

Adalimumab dose escalation is effective for managing secondary loss of response in Crohn's disease - Ma - 2014 - Alimentary Pharmacology & Therapeutics - Wiley Online Library

Prediction of Successful Dose Reduction or Discontinuation of Adalimumab or Etanercept Using Serum Drug Levels and Antidrug Antibody Measurement - ACR Meeting Abstracts

Efficacy, safety and medication cost implications of adalimumab 40 mg weekly dosing in patients with psoriasis with suboptimal response to 40 mg every other week dosing: results from an open‐label study -

Anti-TNF dose escalation and drug sustainability in Crohn's disease: Data from the nationwide administrative database in Hungary - Digestive and Liver Disease


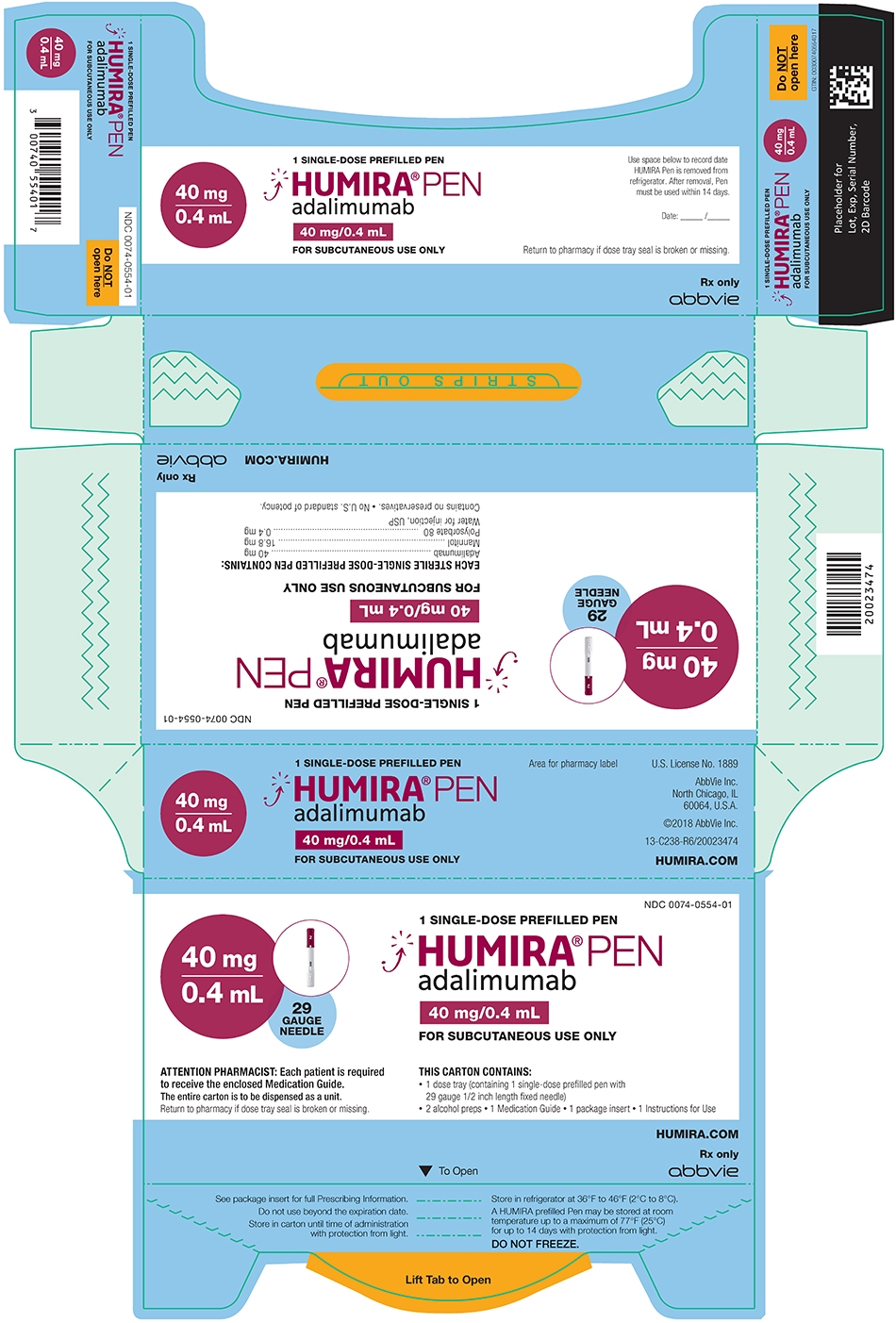




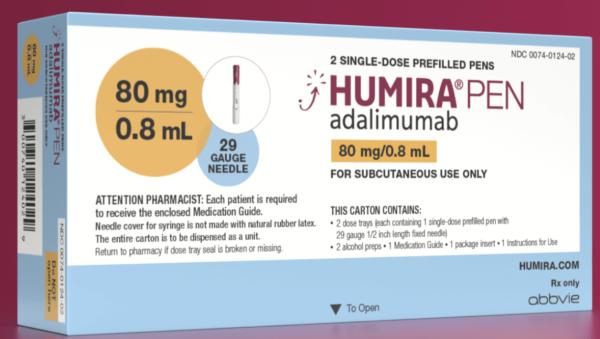






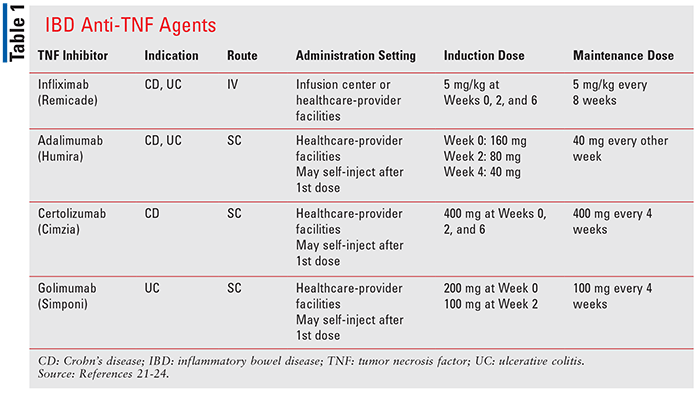
/VWH_Illustration_Drug_Humira_Adalimumab_Illustrator_Dennis-Madamba-d925e94c1115461684f4b3d6ef473ec7.jpg)