Lorlatinib Salvages CNS Relapse in an ALK-Positive Non–Small-Cell Lung Cancer Patient Previously Treated With Crizotinib and High-Dose Brigatinib - Clinical Lung Cancer
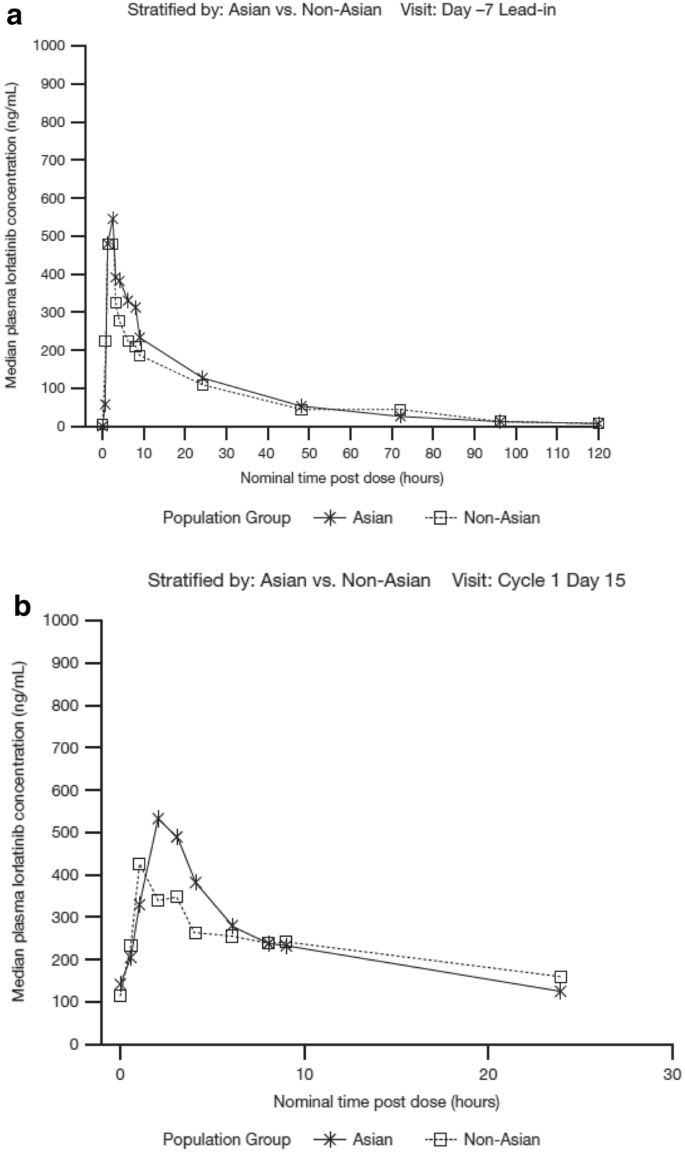
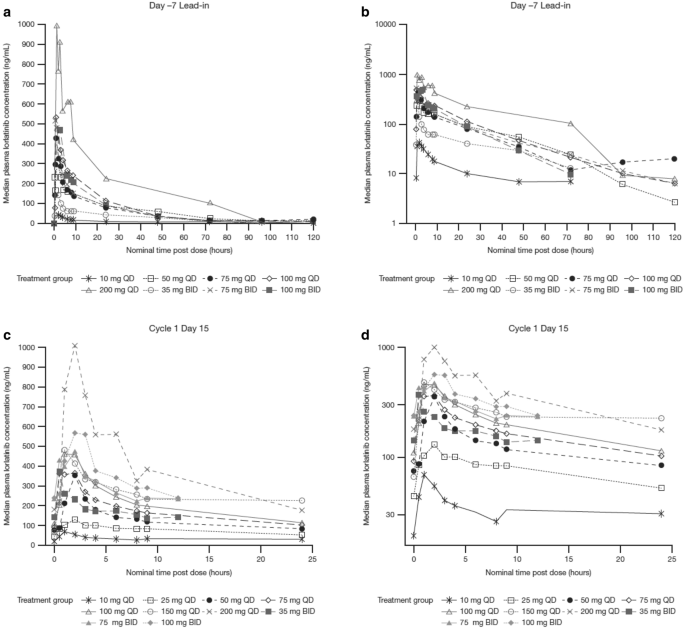
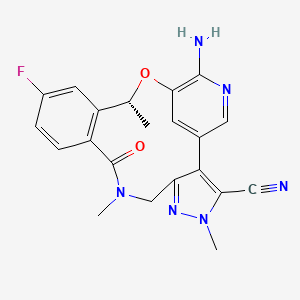
Pharmacokinetics of Lorlatinib After Single and Multiple Dosing in Patients with Anaplastic Lymphoma Kinase (ALK)-Positive Non-Small Cell Lung Cancer: Results from a Global Phase I/II Study | SpringerLink

Continuation of Lorlatinib in ALK-Positive NSCLC Beyond Progressive Disease - Journal of Thoracic Oncology
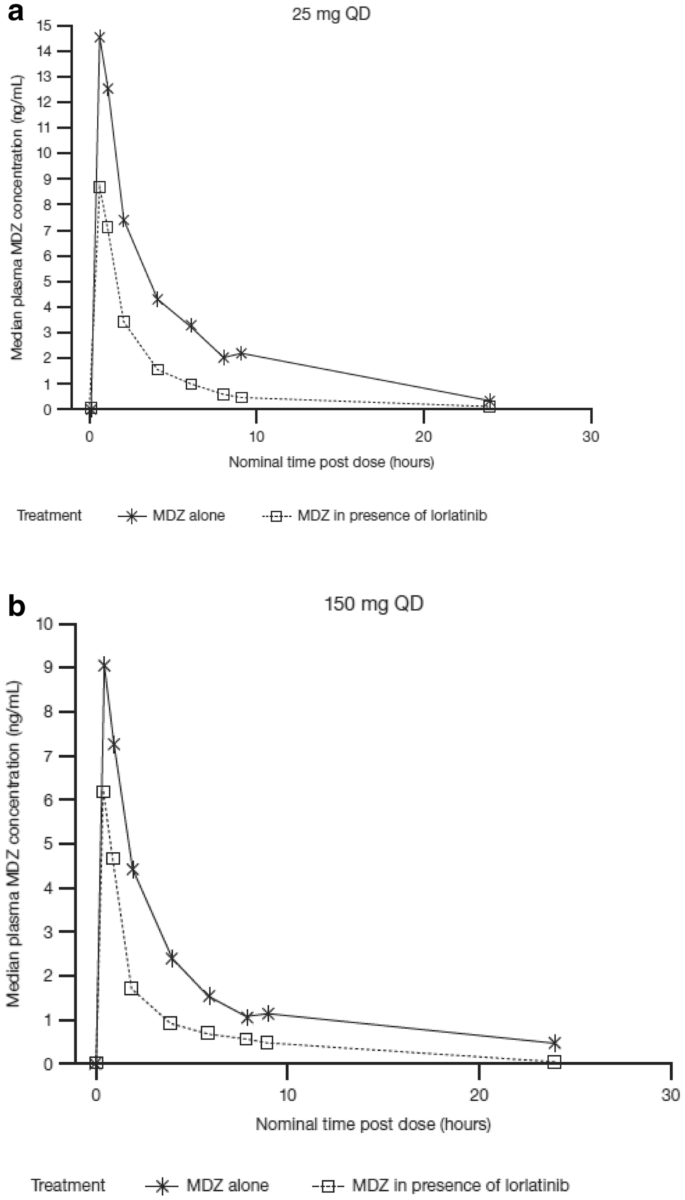
Pharmacokinetics of Lorlatinib After Single and Multiple Dosing in Patients with Anaplastic Lymphoma Kinase (ALK)-Positive Non-Small Cell Lung Cancer: Results from a Global Phase I/II Study | SpringerLink

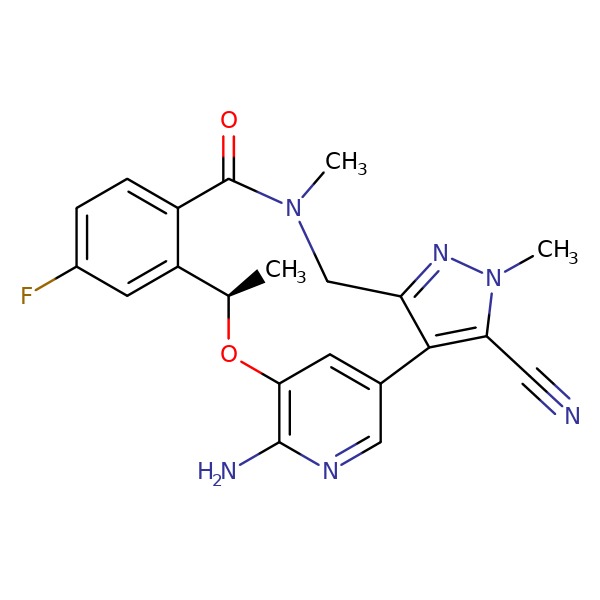
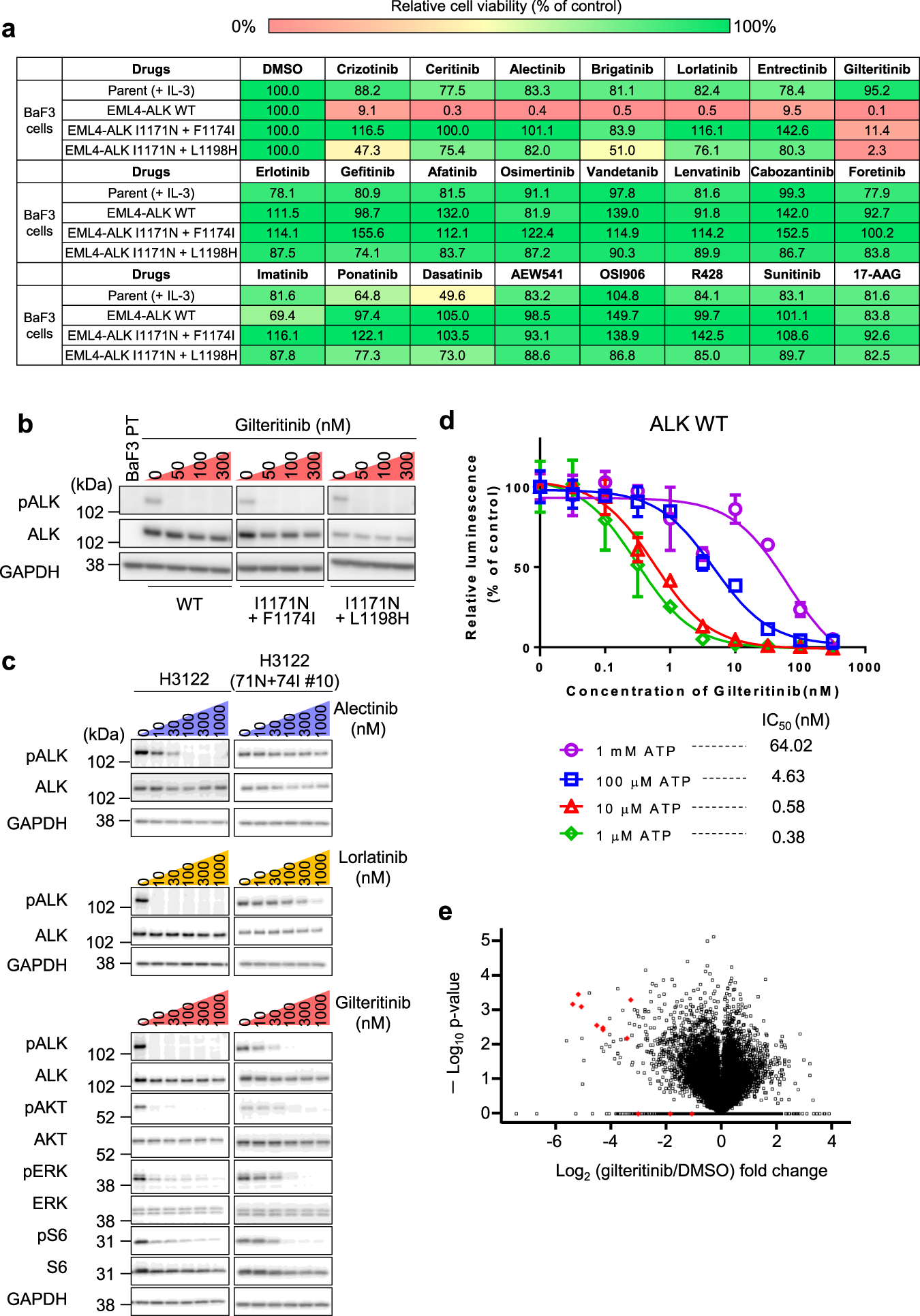
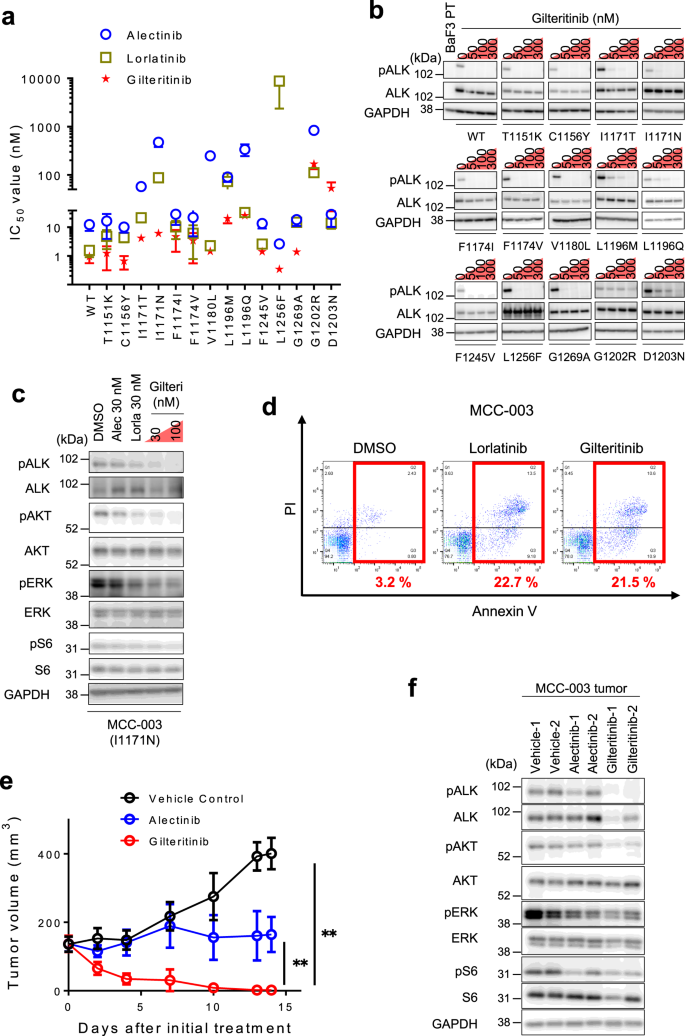
Analysis of lorlatinib analogs reveals a roadmap for targeting diverse compound resistance mutations in ALK-positive lung cancer | Nature Cancer

a Plasma lorlatinib and b metabolite (M8) concentration-time profiles... | Download Scientific Diagram

Lorlatinib in non-small-cell lung cancer with ALK or ROS1 rearrangement: an international, multicentre, open-label, single-arm first-in-man phase 1 trial - The Lancet Oncology

Summary of Plasma Lorlatinib Pharmacokinetic Parameters Following a... | Download Scientific Diagram

Pharmaceuticals | Free Full-Text | Later-Line Treatment with Lorlatinib in ALK- and ROS1-Rearrangement-Positive NSCLC: A Retrospective, Multicenter Analysis | HTML

Phase II Study of Lorlatinib in Patients With Anaplastic Lymphoma Kinase–Positive Lung Cancer and CNS-Specific Relapse | JCO Precision Oncology

Evaluation of the absolute oral bioavailability of the anaplastic lymphoma kinase/c-ROS oncogene 1 kinase inhibitor lorlatinib in healthy participants | SpringerLink