
Synthesis of Natural Rubrolides B, I, K, L, M, O and Analogues - Vries - 2021 - European Journal of Organic Chemistry - Wiley Online Library

Alkyne Aminopalladation/Heck and Suzuki Cascades: An Approach to Tetrasubstituted Enamines - Geffers - 2021 - Chemistry – A European Journal - Wiley Online Library

Isolation of an N‐Heterocyclic Carbene Complex of a Borasilene - Franz - 2019 - Chemistry – A European Journal - Wiley Online Library

Gold-Catalyzed Nucleophilic Cyclization of Functionalized Allenes: A Powerful Access to Carbo- and Heterocycles | Chemical Reviews

Preparation of Quaternary Centers via Nickel-Catalyzed Suzuki–Miyaura Cross-Coupling of Tertiary Sulfones | Journal of the American Chemical Society
Dual infection system identifies a crucial role for PKAâ•'mediated serine phosphorylation of the EPECâ•'Tirâ•'inj
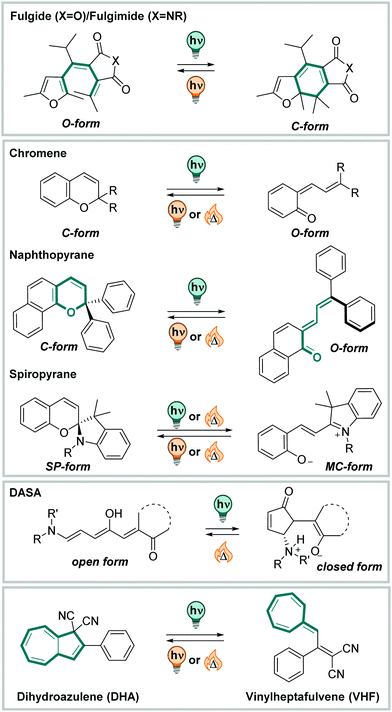
Molecular photoswitches in aqueous environments - Chemical Society Reviews (RSC Publishing) DOI:10.1039/D0CS00547A

Reaction of Azides with Dichloroindium Hydride: Very Mild Production of Amines and Pyrrolidin-2-imines through Possible Indium−Aminyl Radicals | Organic Letters

Palladium‐Catalyzed Suzuki Coupling of N‐Acyloxazolidinones via Selective Cleavage of C–N Bonds - Jian - 2020 - European Journal of Organic Chemistry - Wiley Online Library

Synthesis of Natural Rubrolides B, I, K, L, M, O and Analogues - Vries - 2021 - European Journal of Organic Chemistry - Wiley Online Library
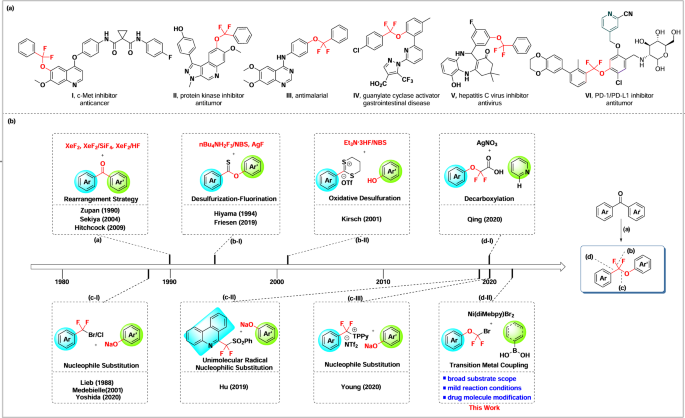
Synthesis of aryldifluoromethyl aryl ethers via nickel-catalyzed suzuki cross-coupling between aryloxydifluoromethyl bromides and boronic acids | Communications Chemistry

Synthesis of Natural Rubrolides B, I, K, L, M, O and Analogues - Vries - 2021 - European Journal of Organic Chemistry - Wiley Online Library









