Reactogenicity and immunogenicity after a late second dose or a third dose of ChAdOx1 nCoV-19 in the UK: a substudy of two randomised controlled trials (COV001 and COV002) - The Lancet

Safety and immunogenicity of the ChAdOx1 nCoV-19 (AZD1222) vaccine against SARS-CoV-2 in people living with and without HIV in South Africa: an interim analysis of a randomised, double-blind, placebo-controlled, phase 1B/2A trial -

Reactogenicity and immunogenicity after a late second dose or a third dose of ChAdOx1 nCoV-19 in the UK: a substudy of two randomised controlled trials (COV001 and COV002) - The Lancet
Apoptosis-Related Gene and Protein Expression in Human Lymphoma Xenografts (Raji) After Low Dose Rate Radiation Using 67Cu-2IT-B

Predicting late radiation-induced xerostomia with parotid gland PET biomarkers and dose metrics - Radiotherapy and Oncology
Deep Learning–based Reconstruction for Lower-Dose Pediatric CT: Technical Principles, Image Characteristics, and Clinical Impl

Landscape and representative microhabitat dose rates at Kamitashiro,... | Download Scientific Diagram

G Live on Twitter: "Vaccinations Update: 18-39 yr olds can walk-in & get the Pfizer vaccine @GLiveGuildford until 7pm today. Pfizer also available for 18-39 yr olds on Fri 8am-7pm & Sat
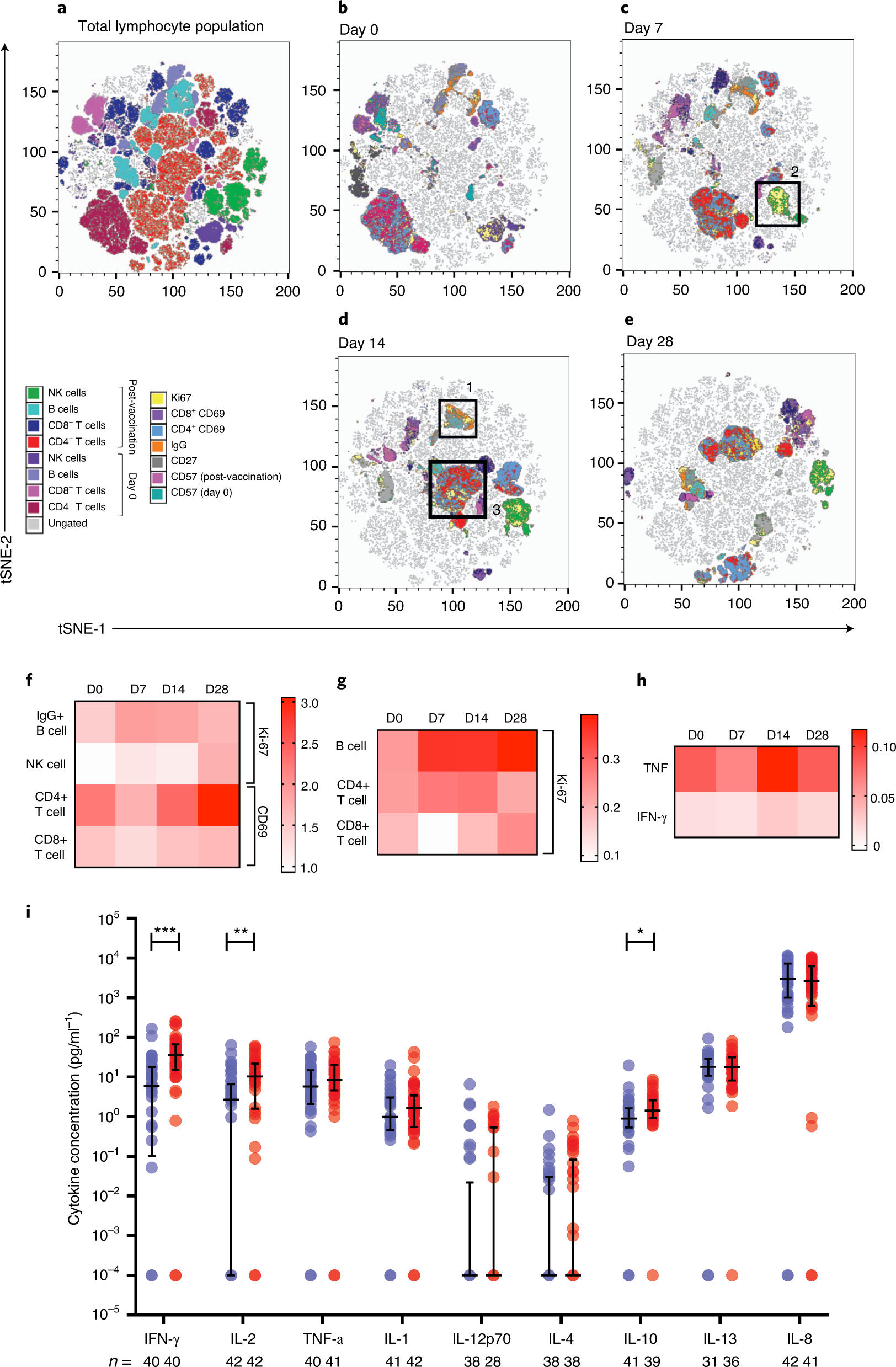
T cell and antibody responses induced by a single dose of ChAdOx1 nCoV-19 (AZD1222) vaccine in a phase 1/2 clinical trial | Nature Medicine

Persistence of immunogenicity after seven COVID-19 vaccines given as third dose boosters following two doses of ChAdOx1 nCov-19 or BNT162b2 in the UK: Three month analyses of the COV-BOOST trial. - Journal
Deep Learning–based Reconstruction for Lower-Dose Pediatric CT: Technical Principles, Image Characteristics, and Clinical Impl
The role of sulphate conjugation in the metabolism and disposition of oral and intravenous paracetamol in man.

Safety and immunogenicity of seven COVID-19 vaccines as a third dose (booster) following two doses of ChAdOx1 nCov-19 or BNT162b2 in the UK (COV-BOOST): a blinded, multicentre, randomised, controlled, phase 2 trial -

Single-dose administration and the influence of the timing of the booster dose on immunogenicity and efficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine: a pooled analysis of four randomised trials - The Lancet

Efficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine against SARS-CoV-2 lineages circulating in Brazil | Nature Communications